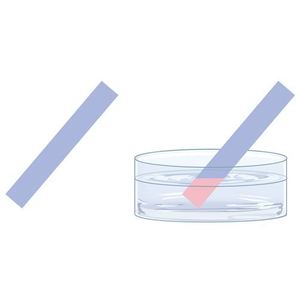
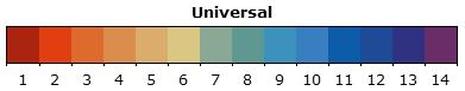
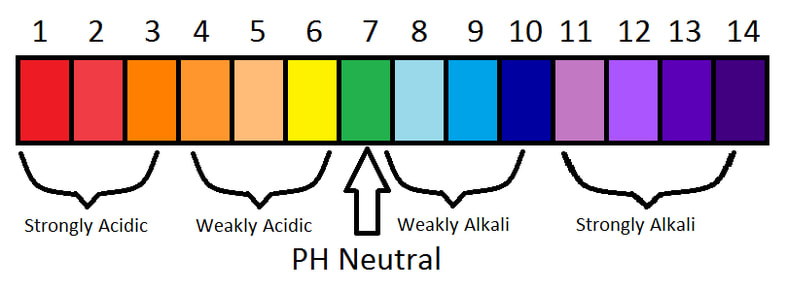
How to Measure Acidity/Alkalinity of a Solution | Acids Vs Bases
|
What are Acids?
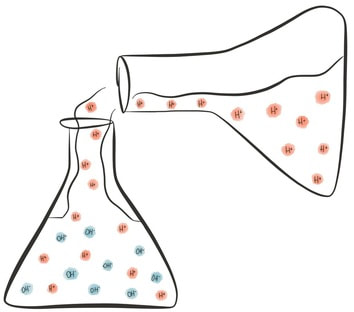
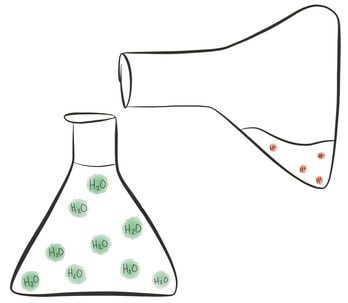
Acids are a group of chemicals that have particular characteristics. All acids contain hydrogen atoms, and they release hydrogen ions (H+) when they dissolve in water, or when they are mixed with a base! Acids can be mineral (man-made, e.g. sulfuric acid in the lab) or organic (found naturally, e.g. citric acid in lemons).
|
What are Bases?
Bases are a group of chemical that are also known as 'acid opposites' - they cancel out the effect of acids by removing the hydrogen ions from a solution. Soluble bases (can dissolve in water) have a special name - they are called alkalis, and when dissolved they release hydroxide ions (OH-).
|
Acid and Base Concentrations
|
ACIDS
The more hydrogen ions (H+) present in the solution, the stronger the acid. We call a strong acid a 'concentrated' acid. If an acid solution doesn't have many H+ ions in it, the acid is weak. We call this a 'dilute' acid.
|
BASES
The more hydroxide ions (OH-) present in the solution, the stronger the alkali (remember bases that can dissolve in water (thus forming a solution) are called alkalis). A strong alkali can be described as a 'concentrated' alkali. If an alkaline solution does not contain many OH- ions in it, the alkali is weak. We call this a 'dilute' alkali.
|